Mùthaidhean Gine 29 EGFR Daonna
Ainm an toraidh
Pasgan Lorgaidh Mùthaidhean Gine 29 EGFR Daonna HWTS-TM0012A (PCR Fluorescence)
Eipeidimeo-eòlas
Tha aillse sgamhain air a bhith mar phrìomh adhbhar bàsan aillse air feadh an t-saoghail, a’ bagairt slàinte dhaoine gu mòr. Tha aillse sgamhain neo-bheag-cheallach cunntachail airson mu 80% de dh’euslaintich aillse sgamhain. An-dràsta, is e EGFR an targaid mhóileciuil as cudromaiche airson làimhseachadh aillse sgamhain neo-bheag-cheallach. Faodaidh fosfaireachadh EGFR fàs cealla tumhair, eadar-dhealachadh, ionnsaigh, metastasis, anti-apoptosis, agus angiogenesis tumhair adhartachadh. Faodaidh luchd-bacadh tyrosine kinase EGFR (TKI) slighe chomharran EGFR a bhacadh le bhith a’ cur bacadh air autofosfaireachadh EGFR, agus mar sin a’ cur bacadh air iomadachadh agus eadar-dhealachadh cheallan tumhair, a’ brosnachadh apoptosis cealla tumhair, a’ lughdachadh angiogenesis tumhair, msaa., gus leigheas targaideach tumhair a choileanadh. Tha àireamh mhòr de sgrùdaidhean air sealltainn gu bheil èifeachdas leigheasach EGFR-TKI ceangailte gu dlùth ri inbhe mùthadh gine EGFR, agus faodaidh e gu sònraichte casg a chuir air fàs cheallan tumhair le mùthadh gine EGFR. Tha an gine EGFR suidhichte air a’ ghàirdean ghoirid de chromosome 7 (7p12), le fad slàn de 200Kb agus tha 28 exons ann. Tha an roinn mùthaichte suidhichte sa mhòr-chuid ann an exons 18 gu 21, tha mùthadh cuir às do chodons 746 gu 753 air exon 19 a’ dèanamh suas mu 45% agus tha mùthadh L858R air exon 21 a’ dèanamh suas mu 40% gu 45%. Tha Stiùiridhean NCCN airson Breithneachadh agus Làimhseachadh Ailse Sgamhain Neo-Bheag-cheallan ag ràdh gu soilleir gu bheil feum air deuchainn mùthaidh gine EGFR mus tèid EGFR-TKI a thoirt seachad. Tha an inneal deuchainn seo air a chleachdadh gus stiùireadh a thoirt do rianachd dhrogaichean casg tyrosine kinase gabhadair factar fàis epidermal (EGFR-TKI), agus gus am bunait a thoirt seachad airson leigheas pearsanaichte dha euslaintich le aillse sgamhain neo-bheag-cheallan. Chan eil an inneal seo air a chleachdadh ach airson lorg mùthaidhean cumanta anns a’ ghine EGFR ann an euslaintich le aillse sgamhain neo-bheag-cheallan. Tha na toraidhean deuchainn airson fiosrachadh clionaigeach a-mhàin agus cha bu chòir an cleachdadh mar an aon bhunait airson làimhseachadh pearsanaichte euslaintich. Bu chòir do luchd-clionaigeach beachdachadh air suidheachadh an euslaintich, comharran dhrogaichean, agus làimhseachadh. Tha an ath-bhualadh agus comharran deuchainn obair-lann eile agus factaran eile air an cleachdadh gus breithneachadh coileanta a dhèanamh air na toraidhean deuchainn.
Sianal
| FAM | Bufair Ath-bhualadh IC, Bufair Ath-bhualadh L858R, Bufair Ath-bhualadh 19del, Bufair Ath-bhualadh T790M, Bufair Ath-bhualadh G719X, Bufair Ath-bhualadh 3Ins20, Bufair Ath-bhualadh L861Q, Bufair Ath-bhualadh S768I |
Paramadairean Teicnigeach
| Stòradh | Lionn: ≤-18℃ Anns an dorchadas; Liophilichte: ≤30℃ Anns an dorchadas |
| Beatha sgeilp | Leaghan: 9 mìosan; Lyophilisichte: 12 mìosan |
| Seòrsa Eisimpleir | clò ùr tumhair, earrann pathological reòta, clò no earrann pathological leabaithe ann am paraffin, plasma no serum |
| CV | <5.0% |
| LoD | lorg fuasgladh ath-bhualadh searbhag niùclasach fo chùl-raon seòrsa fiadhaich 3ng / μL, faodaidh e ìre mùthaidh 1% a lorg gu seasmhach |
| Sònrachas | Chan eil crois-imoibriadh ann le DNA genomic daonna fiadhaich agus seòrsachan mutant eile. |
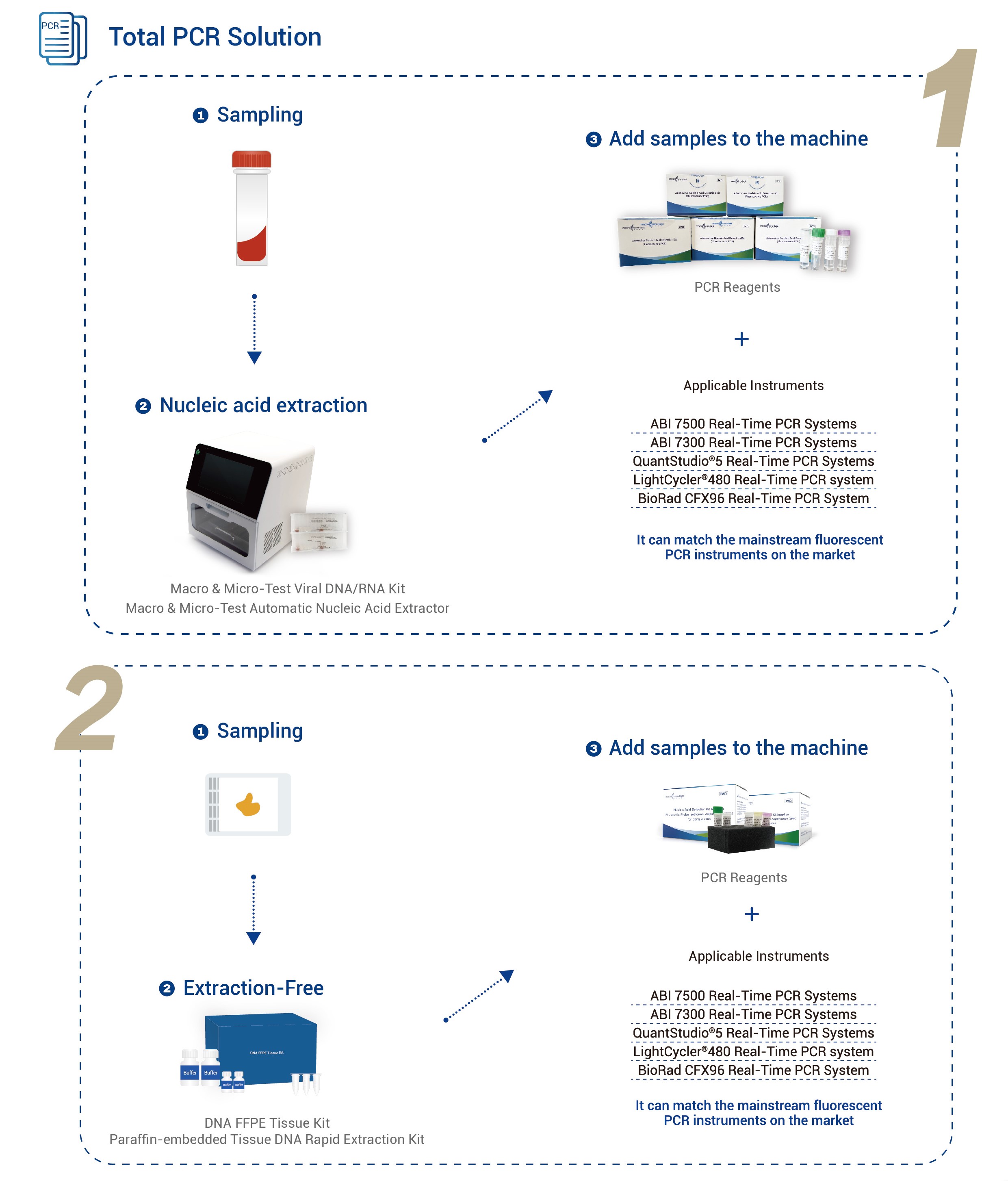
| Ionnsramaidean Iomchaidh | Siostaman PCR Fìor-Ùine Applied Biosystems 7500Siostaman PCR Fìor-Ùine Applied Biosystems 7300 Siostaman PCR Fìor-Ùine QuantStudio® 5 Siostam PCR Fìor-Ùine LightCycler® 480 Siostam PCR Fìor-ùine BioRad CFX96 |